Helpful Resources
American Thyroid Association Guidelines
The Guidelines were compiled by American Thyroid Association® Guidelines Taskforces selected for their expertise on the topic. The Guidelines are reviewed by the ATA® Board of Directors and submitted to the ATA® membership for comment.
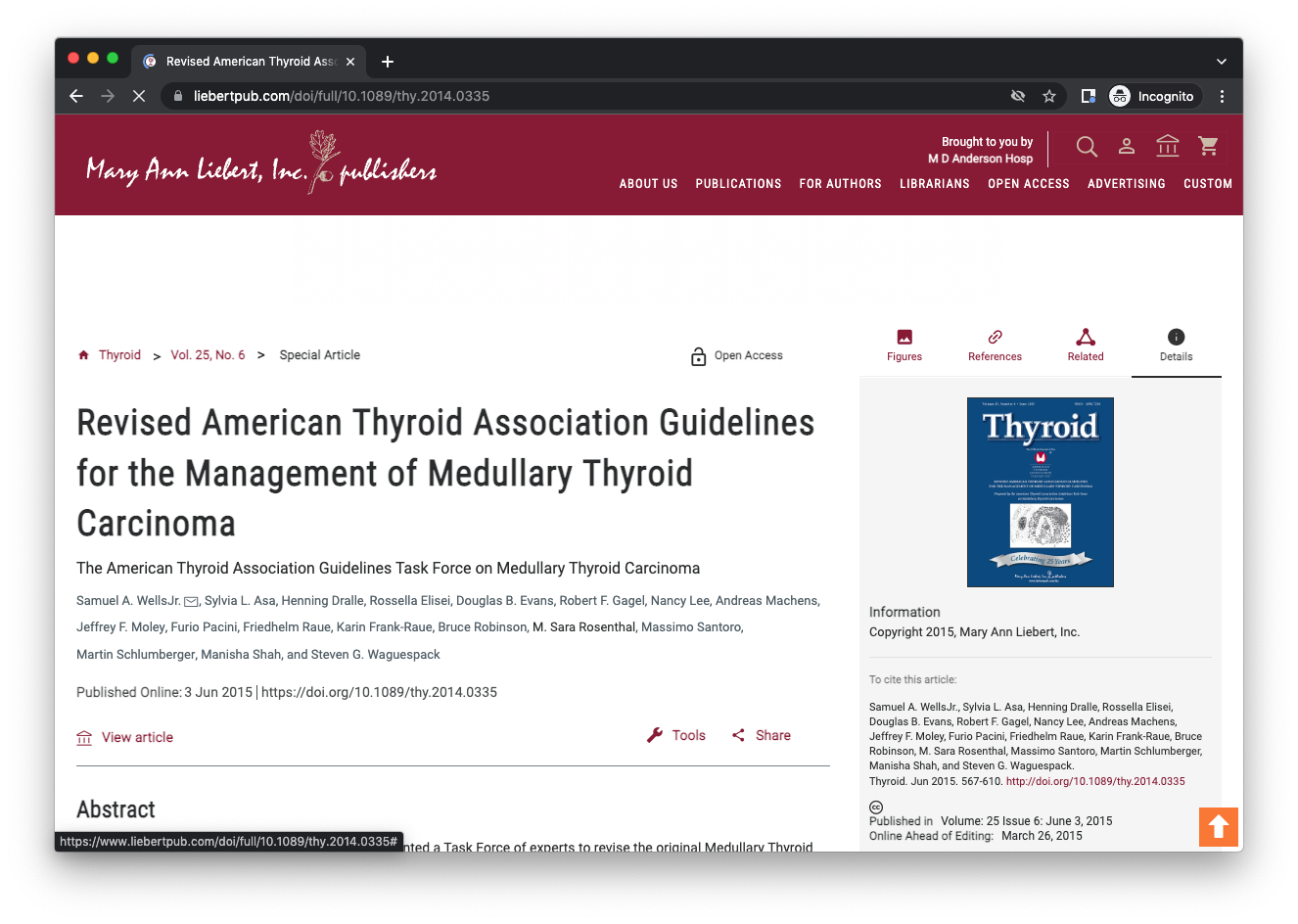
Revised American Thyroid Association Guidelines for the Management of Medullary Thyroid Carcinoma
The American Thyroid Association appointed a Task Force of experts to revise the original Medullary Thyroid Carcinoma: Management Guidelines of the American Thyroid Association.
The Task Force developed 67 evidence-based recommendations to assist clinicians in the care of patients with MTC. The Task Force considers the recommendations to represent current, rational, and optimal medical practice.
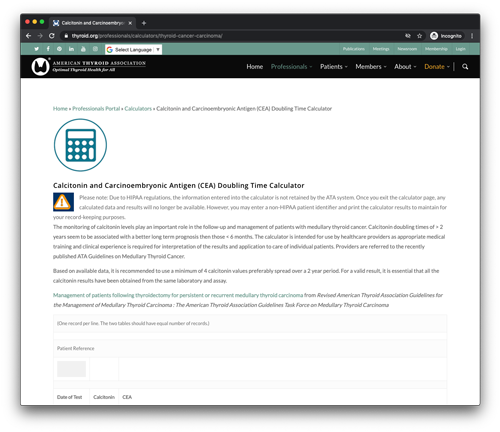
American Thyroid Association: Calcitonin and Carcinoembryonic Antigen (CEA) Doubling Time Calculator
The monitoring of calcitonin levels play an important role in the follow-up and management of patients with medullary thyroid cancer. Calcitonin doubling times of > 2 years seem to be associated with a better long term prognosis then those < 6 months. The calculator is intended for use by healthcare providers as appropriate medical training and clinical experience is required for interpretation of the results and application to care of individual patients. Providers are referred to the recently published ATA Guidelines on Medullary Thyroid Cancer.
Based on available data, it is recommended to use a minimum of 4 calcitonin values preferably spread over a 2 year period. For a valid result, it is essential that all the calcitonin results have been obtained from the same laboratory and assay.
Support Groups
ThyCa: Thyroid Cancer Survivors’ Association, Inc.
ThyCa: Thyroid Cancer Survivors’ Association, Inc. is a non-profit 501(c)(3) organization (tax ID #52-2169434) of thyroid cancer survivors, family members, and health care professionals. We are dedicated to support, education, and communication for thyroid cancer survivors, their families and friends. We also sponsor Thyroid Cancer Awareness Month, year-round awareness activities, and thyroid cancer research fundraising and research grants.
AMEND: Association for Multiple Endocrine Neoplasia Disorders
AMEND is a registered UK non-profit that provides information and support services to anyone affected by multiple endocrine neoplasia (MEN) disorders and associated endocrine tumours, including MTC. Members benefit from a free psychological counselling service, patient information resources and support services and events. We award annual research grants when funds allow. Membership is free and open to all via our website at www.amend.org.uk
AMEND USA: Association for Multiple Endocrine Neoplasia USA
AMEND USA is a US-based non-profit connecting the MEN disorders community–patients, family, friends, and providers–with useful resources, including on MTC. Please see our website for more information, to join our mailing list, and contact information.
AMENSupport: American Multiple Endocrine Neoplasia Support
A diagnosis of Multiple Endocrine Neoplasia (MEN) often brings with it more questions than you know how to ask. Chances are, the path to this diagnosis has been a long road with much uncertainty. This website is designed to get you started on the road to learning about your rare disease and to offer you resources to help along the way.
Opportunities to Participate in Research
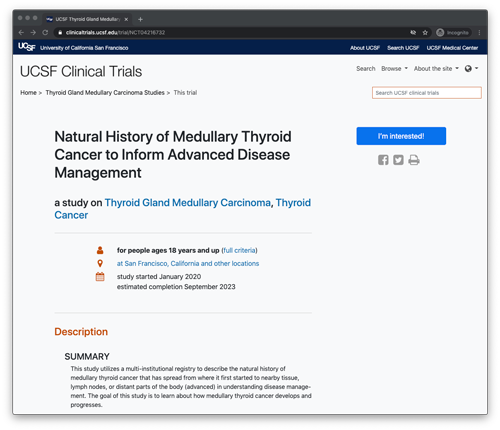
MD Anderson Cancer Center /University of California San Francisco Registry Information
The MTCR is comprised of both types of MTC, sporadic disease, which is MTC not passed down within families, and hereditary disease, which is inherited. The type of MTC you have will determine which version of the registry you will be enrolled.
Study for adult Medullary Thyroid Cancer Patients Starting New Treatment
The goal of this research study is to describe the natural history of MTC using the existing MTC Registry in order to understand the pathway to progressive disease requiring drug therapy. In particular, this study plans to use a brief, daily survey and measure of blood pressure to see if this more frequent monitoring can provide information to improve care of current treatment and help shape future clinical trials. We are interested in enrolling individuals who are at least 18-years old, have a diagnosis of medullary thyroid cancer, are starting a new standard therapy or clinical trial for the treatment of MTC, and who have been previously enrolled in the MTC Registry.
ClinicalTrials.gov
The ClinicalTrials.gov Web site provides current information about clinical research studies to patients, their families and caregivers, health care professionals, and the public. Each study record includes a summary of the study protocol, including the purpose, recruitment status, and eligibility criteria. Study locations and specific contact information are listed to assist with enrollment. (See How to Read a Study Record to learn more about the information found in a study record.) Information on ClinicalTrials.gov is provided and updated by the sponsor or principal investigator of the clinical study. Clinicaltrials.gov is a free service of the National Institutes of Health (NIH) and is maintained by the National Library of Medicine (NLM).
Current Studies Available to Join at MDA/UCSF

MTC Registry Contact Information
- MD Anderson Cancer Center: genr@mdanderson.org (713-563-2879)
- UCSF: MTCRegistry@ucsf.edu (415-476-2816)

Study for adult Medullary Thyroid Cancer Patients (MTC Registry)
This 3 year study aims to analyze the natural history of Medullary Thyroid Carcinoma. It will utilize information that is being collected from participant medical records in the MTC Registry and the patient-reported outcome data from the questionnaires that are part of the Registry. This study is funded by the U.S. Food and Drug Administration and participants will be compensated with a $10 gift card for each series of questionnaires they complete at each time point. We are inviting people who are at least 18 years-old, have a diagnosis of medullary thyroid cancer, and are a participant in the Sporadic or Hereditary Registry.
Financial Resources
Finding and Paying for Treatment (American Cancer Society)
Financial Assistance (CancerCare)
A Helping Hand: The Resource Guide for People With Cancer by CancerCare (Publication Date: 2021)
This comprehensive booklet includes national and regional organizations that offer various kinds of financial help to people with cancer. The listings include contact information for each organization and a description of the type of help provided. You can also see categories that tell you, at a glance, the type of help available.
Health Reform Glossary
Learn more about health care industry terms from the Kaiser Foundation
Financial and Legal Assistance Recommended Resources
Use this guide from the MD Anderson Cancer Center-Learning Center to learn more about financial and legal topics related to cancer care and survivorship.
MD Anderson Cancer Center: Insurance, Billing and Financial Support
Paying your hospital bill and worrying about insurance claims may be the last thing you or your loved ones want to deal with while you are coping with cancer. That’s why MD Anderson has financial specialists committed to helping you throughout your care.
MD Anderson’s Patient Business Services teams and Financial Clearance Center work behind the scenes to help you understand the insurance and billing process so you can focus on your cancer treatment.
Our customer service representatives are here to answer questions about your bill and patient statement. You may reach us online through MyChart.
American Cancer Society: Managing the Costs of Cancer Treatment
Cancer care and treatment can be costly. Here are some tips on what costs you might be able to expect and some ideas on how to plan for, ask about, and discuss treatment costs with your cancer care team.
Cancer Financial Assistance Coalition
CFAC is a coalition of financial assistance organizations joining forces to help cancer patients experience better health and well-being by limiting financial challenges, through:
- Facilitating communication and collaboration among member organizations;
- Educating patients and providers about existing resources and linking to other organizations that can disseminate information about the collective resources of the member organizations;
- Advocating on behalf of cancer patients who continue to bear financial burdens associated with the costs of cancer treatment and care.
Medicine Assistance Tool
Patient Advocate Foundation
Patient Advocate Foundation (PAF) is a national 501 (c)(3) non-profit organization which provides case management services and financial aid to Americans with chronic, life threatening and debilitating illnesses.
Patient Advocate Foundation: Co-Pay Relief
Patient Advocate Foundation: Financial Aid Funds
Eligibility is different for each specific fund, refer to the program details to identify if criteria is met. The purpose of each fund is defined in the fund details.








 So far I have learned that there is no room for second-guessing. Go forth and gather the best information to make the best decisions (regarding treatment) and move on. Do not look back. And never ask yourself, "Should I have...?" or "What if?"
So far I have learned that there is no room for second-guessing. Go forth and gather the best information to make the best decisions (regarding treatment) and move on. Do not look back. And never ask yourself, "Should I have...?" or "What if?"















